New insights into rare pancreatic tumors that cause low blood sugar
Researchers identify a key gene involved in abnormal insulin secretion in insulinomas, offering hope for new diagnostics and therapeutic strategies
The dedicator of cytokinesis 10 (DOCK10) gene has been identified as a key driver of abnormal insulin secretion in insulinomas, as reported by researchers from Institute of Science Tokyo. Using surgical specimens and patient-derived organoids, the team performed comprehensive genetic and transcriptomic analyses, revealing that inhibiting a DOCK10-related pathway reduced excessive insulin release in cellular and animal models. These results pave the way for novel diagnostic biomarkers and treatment options for insulinomas.
DOCK10 in Insulin Hypersecretion in Human Insulinoma
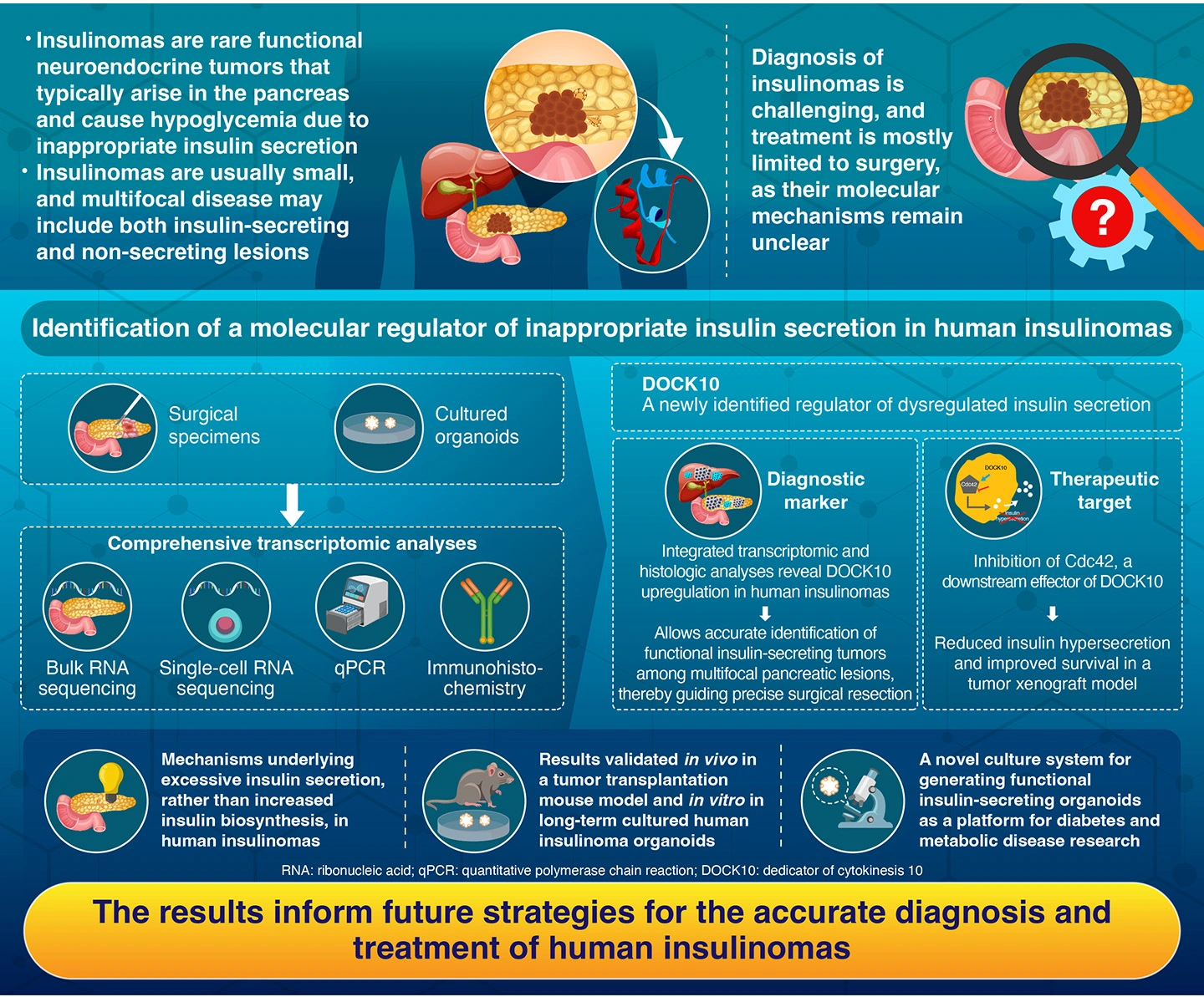
Insulinomas are rare tumors of the pancreas that release insulin in excess. Even small tumors are often dangerous, as they can trigger severe drops in blood sugar (hypoglycemia), leading to dizziness, headaches, fatigue, and even seizures, among other symptoms. In most cases, surgery is the primary and most effective treatment for this type of tumor.
However, diagnosing and managing insulinomas is not always straightforward. In some patients, especially those with metastatic disease, multiple tumors can be present at the same time, some of which secrete insulin while others do not. Although imaging and insulin staining can confirm that excess insulin is being produced, doctors cannot reliably determine which tumor or tumors are responsible for hypoglycemia. A major obstacle to developing better diagnostic techniques and treatments for insulinomas has been our limited understanding of the molecular mechanism behind their abnormal insulin secretion.
To address this challenge, a research team led by Assistant Professor Go Ito from the Department of Gastroenterology and Hepatology, the Center for Personalized Medicine for Healthy Aging, Institute of Science Tokyo (Science Tokyo), Japan, and graduate student Hiromune Katsuda from the Department of Gastroenterology and Hepatology, Graduate School of Medical and Dental Sciences, Science Tokyo, in collaboration with Professor Philip Rosenstiel from the Institute of Clinical Molecular Biology, Christian-Albrechts-University and University Hospital Schleswig-Holstein, Germany, conducted an in-depth investigation into the biology of insulinomas. As explained in their paper, the team combined analyses of surgical tumor specimens with patient-derived organoids—miniature lab-grown versions of tumors—to get a clearer picture of what drives abnormal insulin release. The study was published online in Volume 20, Issue 5 of the journal Cellular and Molecular Gastroenterology and Hepatology on December 11, 2025.
The researchers first established a biobank of insulinoma samples and cultured organoids produced from patients’ tissues. Then, they performed large-scale gene expression analyses, including bulk RNA sequencing, single-cell RNA sequencing, quantitative PCR, and immunohistochemistry. These techniques revealed that insulinomas exhibited marked changes in genes involved in the insulin secretion machinery, rather than in insulin production itself. Simply put, the tumors were not making more insulin but releasing it inappropriately.
Among the genes identified, dedicator of cytokinesis 10 (DOCK10) stood out the most. The researchers found that this gene was strongly and specifically expressed in insulin-secreting tumor cells but not in normal pancreatic tissue or other types of pancreatic tumors. Further experiments revealed that DOCK10 activates a signaling pathway involving Cdc42, a protein that helps regulate insulin exteriorization (exocytosis). When this pathway was blocked using a Cdc42 inhibitor, insulin hypersecretion decreased in insulinoma cells, in human insulinoma organoids, and in tumor models transplanted into mice. These mice also showed improved survival. "Our results suggest that DOCK10 may serve as a diagnostic marker for insulin-secreting lesions and a potential therapeutic target in insulinoma. It provides mechanistic insights that could inform future strategies for precision diagnostics and treatment of pancreatic neuroendocrine tumors," explains Ito.
Another remarkable achievement of this study was the successful development of a long-term culture system for human insulinoma organoids. "We observed consistent gene expression profiles between insulinoma tissues and corresponding organoids, and insulin secretion over an extended period was also confirmed. These findings suggest that our culture system successfully retains key functional properties of the original tumors," remarks Ito. "The sustained culture of insulin-secreting human cells using our approach may offer opportunities for disease modeling and therapeutic testing. As culture systems continue to improve, insulinoma-derived cells could even be explored as a potential source for transplantation therapies."
Overall, this work sheds light on the unique biological mechanisms found in insulinomas, paving the way for more accurate diagnostic techniques and targeted treatments for these rare pancreatic tumors.
Reference
- Authors:
- Hiromune Katsuda1, Go Ito2,*, Franziska Kimmig3, Tomohiro Muto1, Neha Mishra3, Joana Pimenta Bernardes3, Yui Hiraguri1, Hironari Yamashita4, Akira Ito4, Yuko Kinowaki5, Takahiro Shin1, Satoru Fujii1, Masato Miyoshi1, Masanori Kobayashi1, Daisuke Asano4, Yoshiya Ishikawa4, Hiroki Ueda4, Keiichi Akahoshi4, Eriko Katsuta4, Yoshihito Kano1,6, Shiro Yui7, Yasuhiro Nemoto1, Atsushi Kudo8, Daisuke Ban4, Yasuhiro Asahina1, Stefan Schreiber3,9, Mamoru Watanabe10, Philip Rosenstiel3, and Ryuichi Okamoto1
- Title:
- DOCK10 Regulates Insulin Hypersecretion in Insulinoma and Serves as a Diagnostic and Therapeutic Target
- Journal:
- Cellular and Molecular Gastroenterology and Hepatology
- Affiliations:
- 1Department of Gastroenterology and Hepatology, Institute of Science Tokyo, Japan
2The Center for Personalized Medicine for Healthy Aging, Institute of Science Tokyo, Japan
3Institute of Clinical Molecular Biology, Christian-Albrechts-University and University Hospital Schleswig-Holstein, Campus Kiel, Germany
4Department of Hepatobiliary and Pancreatic Surgery, Institute of Science Tokyo, Japan
5Department of Comprehensive Pathology, Institute of Science Tokyo, Japan
6Department of Medical Oncology, Institute of Science Tokyo, Japan
7Center for Stem Cell and Regenerative Medicine, Institute of Science Tokyo, Japan
8Department of Clinical Quality and Safety, Institute of Science Tokyo, Japan
9Department of Internal Medicine I, Christian-Albrechts-University and University Hospital Schleswig-Holstein, Campus Kiel, Germany
10Organoid Center, Graduate School of Medicine, Juntendo University, Japan
Related articles
Further information
Assistant Professor Go Ito
The Center for Personalized Medicine for Healthy Aging, Institute of Science Tokyo, Japan
- gitogast@tmd.ac.jp
Contact
Public Relations Division, Institute of Science Tokyo
- media@adm.isct.ac.jp
- Tel
- +81-3-5734-2975