Rethinking intestinal regeneration
How cellular “rejuvenation” helps rebuild the intestine
What the research is about
Our intestines are actually very vulnerable. Every day, they are exposed to damage from food, bacteria, and even medicines. Yet they continue to function properly. This is because our bodies constantly produce new cells that replace damaged ones and take over their functions.
These new cells are generated from stem cells. Stem cells have the unique ability to produce many different types of cells. In the intestine, for example, the inner surface has many small folds, and at the base of these folds are stem cells known as Lgr5-positive stem cells (CBCs). These CBCs have long been thought to be the source of all the different cell types that make up the intestinal lining.
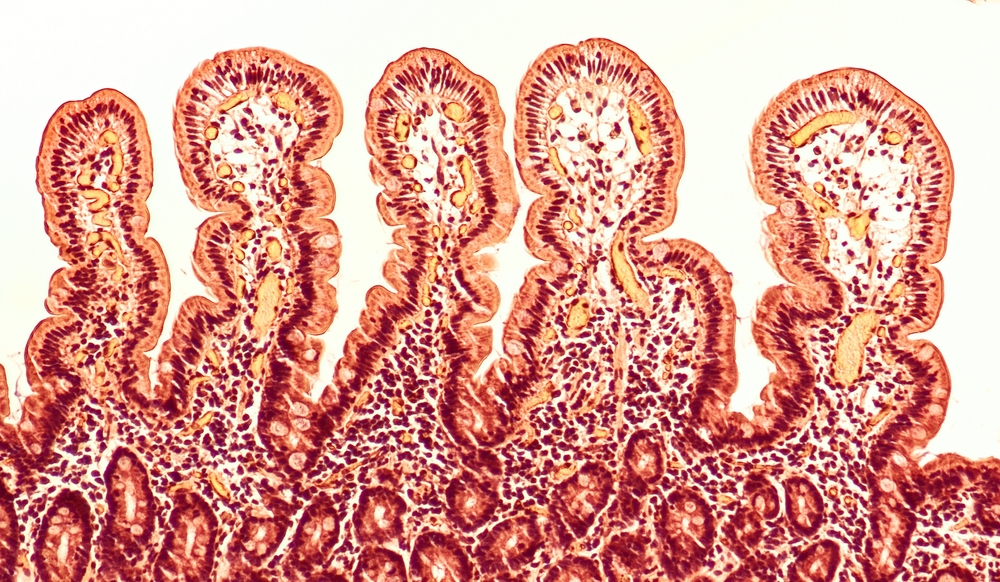
However, when the intestine suffers severe damage—such as from inflammation or chemotherapy—these CBCs can be lost or greatly reduced. In such situations, two remarkable phenomena are known to occur. First, some surviving CBCs revert to a younger, fetal-like state called revival stem cells (revSCs). Second, cells that have already specialized for specific roles—such as intestinal epithelial cells that absorb nutrients—can step in and function in place of stem cells.
Despite these observations, an important question remained unanswered: How are these two phenomena connected during regeneration?
In addition, while it was known that specialized cells could sometimes replace stem cells, the mechanism behind this process was unclear. In particular, it was not understood how differentiated cells—cells that have already acquired specific functions—could regain stem cell-like abilities without undergoing cancer-like changes.
A research team led by Associate Professor Shiro Yui at Institute of Science Tokyo (Science Tokyo) set out to solve this challenging problem by uncovering both the mechanism of intestinal regeneration and the relationships between these cellular processes.
Why this matters
Using advanced techniques such as single-cell analysis—which allows researchers to examine gene activity in individual cells—the team carefully investigated how the intestine regenerates. Their findings revealed several key insights:
- Bidirectional conversion: CBCs and revSCs can convert into each other depending on the situation.
- Epithelial cells can become stem cells: Although intestinal epithelial cells do not normally function as stem cells, they can revert to a fetal-like revSC state and then become CBCs again. Once they become CBCs, they can once again produce various intestinal cell types. In other words, returning to a fetal-like state serves as an important entry point for regeneration.
- A survival strategy under harsh conditions: revSCs are highly resistant to stress such as inflammation and chemotherapy. By reverting to this state, cells can help preserve the stem cell pool.
- Stem cell regeneration without oncogenic changes: Previously, it was believed that non-stem cells could regain stem cell functions when they underwent abnormal changes, such as those seen in cancer. However, this study is the first in the world to show that, under a specific collagen environment, non-stem cells can generate intestinal organoids (miniature intestines) via a fetal-like state—without clear evidence of such mutations. This overturns a long-standing assumption in stem cell biology.
These findings demonstrate that intestinal regeneration is not driven solely by a fixed population of stem cells. Instead, it is supported by a flexible system in which different cells can switch roles and cooperate with one another. Cells are not permanently locked into a single role—they can reset and adapt when needed.
What’s next
The insights gained from this research may contribute to the development of new therapies that promote efficient intestinal repair after injury. They may also help explain why cancer cells are resistant to stress, such as drug treatments.
Furthermore, similar rejuvenation mechanisms may exist in other organs. Understanding these processes could provide important clues about the remarkable resilience and adaptability of living systems.
Comment from the researcher
These findings show how flexible intestinal cells are and how they use sophisticated strategies to survive under harsh conditions. As a physician specializing in gastroenterology, I believe that research on revSCs is also highly important for the advancement of clinical medicine. Returning to a fetal-like state is not simply a regression—it is a proactive strategy to protect the future. By further investigating this cellular ‘escape route,’ we hope to bring new hope to patients suffering from disease.
(Shiro Yui, Associate Professor, Center for Stem Cell and Regenerative Medicine, Institute of Biomedical Engineering, Institute of Science Tokyo / Gastroenterologist, Institute of Science Tokyo Hospital)

Dive deeper
Explore more research in Science for All
Science for All showcases cutting-edge research at Science Tokyo and highlights the ideas, people, and possibilities shaping the future.

Contact
Research Support Service Desk